Why Are Syringe Pumps Needed for Oil and Gas Research?
The accurate volumetric delivery and the flow rate of gases and high viscosity oil samples is very crucial in an analytical experiment.
With the use of modern syringe pump systems, the volumes of both oil and gas samples are correctly known at the final pressure. As a result, there is less concern about the compressibility (degree of volume change) and smooth flow of gas and oil samples respectively. Hence, smart syringe pumps provide a near pulse-free delivery of samples including oil and gas. Therefore, in addition to the routine applications, syringe pumps are preferred for a variety of experimental setups with flow-sensitive detectors including flame based and mass spectrometric detectors1.
Smart syringe pumps are different as compared to the other types of pumps. One of the most significant problems encountered by most of the pumps is an inadequate supply of the viscous liquids to the experimental system. Syringe pumps offer the solution to this problem by providing high-pressure and pressure control for the sample delivery2.
Applications of Syringe Pumps for Oil and Gas: Oil Extraction and Supercritical Fluid
Supercritical fluid1 extraction (SFE) is an important sample preparation technique for the chemical analysis of food3, petrochemical, environmental assays, etc. Syringe pumps play a crucial role in supercritical extraction and analysis of oil samples using supercritical fluids (a dense gas).
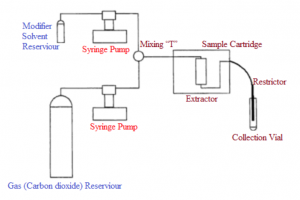
Carbon dioxide is the most popular supercritical solvent2. It is extensively used for the methanolysis or extraction of different oils. At room temperature, the vapor pressure of the carbon dioxide is below its critical pressure. To increase and to control the pressure, a pump is required. The higher the pressure, the higher will be the density and hence the solvent strength of the supercritical fluid. Figure 1 represents the employment of two smart syringe pumps in a typical supercritical fluid system for the delivery of modifier solvent and supercritical gas to an extraction chamber. Different types of pumps including syringe pumps and reciprocating piston pumps have been used for exploring their applications in supercritical fluid extraction techniques. Smart syringe pumps have proved to be a better choice because they offer high precision volumetric delivery, smoother pressure control (i.e., pulse-free delivery) and a better flow rate of the analyte4.
Conclusion
Syringe pumps are a crucial component for sensitive experimental setups for various types of analysis. High-pressure syringe pumps such as the Fusion 6000 are vital for the precise and pulse free delivery of oil and gas samples for a wide range of research and development including green chemistry applications.
References
- Dolan, J. W and, Snyder, L. R. 1989. Troubleshooting LC Systems: A Comprehensive Approach to Troubleshooting LC Equipment and Separations, Springer Science & Business Media
- Nesbitt, B. 2006. Handbook of Pumps and Pumping: Pumping Manual International, Elsevier
- Jackson, M. A and, King, J. W. 1996. Methanolysis of Seed Oils in Flowing Supercritical Carbon Dioxide. JAOCS. 73 (3): 353-356
- Luthria, D. L. 2004. Oil Extraction and Analysis: Critical Issues and Comparative Studies, AOCS Press, USA
- PAC. 1993. Nomenclature for liquid-liquid distribution (solvent extraction) (IUPAC Recommendations 1993) 65(2373): 2381
1Supercritical fluid is a form of matter in which liquid and gaseous phases are indistinguishable. Unlike liquids, a supercritical fluid is a gas that has densities comparable to liquids. However, supercritical fluids are still compressible and expand to completely fill its container. These characteristics enhance the solvating power of the supercritical fluid4.
2Carbon dioxide is commonly found in some food and beverages. It is non-flammable and less toxic gas as compared to many common organic and chlorinated solvents being used for similar applications. All these factors make carbon dioxide a green and attractive supercritical fluid4.
3A modifier is a substance added to a solvent to improve its properties e.g. by increasing the solubility of an extractant, changing interfacial parameters or reducing adsorption losses5.

