Cell analysis can be performed in several ways for example through localizing spectroscopy or with the confocal microscope. However, in the study of large cell populations flows cell cytometry is the best technique to characterize and differentiate cells. Currently, flow cytometry allows the elucidation of bacteria and human cell populations. Nevertheless, it is necessary to have a label to distinguish among cells, in many cases it is possible to apply a tag on target cells. When this labeling is not possible, the intrinsic properties of the cells may be used for that purpose. An unusual technique is an impedance; this electrochemical alternate current method allows the identification of cells because they change their impedance fingerprint. In this summary, you will find key information about the impedance technique and its integration with syringe pumps for cell identification and analysis.
What is impedance and its relationship with syringe pumps?
Impedance is an alternating current technique, widely used in biology but now very well understood. However, this brief review aims to explain the role of syringe pumps in the performance of impedimetric measurements (Figure 1).
Drug therapy and an impedance coupled with syringe pump devices.
It is possible to investigate microfluidic dosage systems for paclitaxel-treated HeLa cells through a Digital syringe pump. The differentiation name is made by impedance spectroscopy; the authors can distinguish between treated and untreated cells using a microfluidic system. Impedance analysis not only allowed the identification is also modeling to establish the theoretical differences between cells. They observed that cell membranes changed and therefore the impedimetric analysis was different for every studied cell. The most astonishing result was the fitting between the experimental results and the theoretical model; which provides an enhanced value of the impedance technique for cell differentiation and future medical applications1.
Furthermore, impedance is cheaper compared to classical tagged techniques like fluorescence or chemical labeling. Cell drug therapy development will favor this kind of study, and this new tool is a promising technology1. The study microfluidic regime was possible with a Nexus 3000 syringe pump (Chemyx Inc., USA) at a rate of 0.4 μL/min. The measurements were carried out with three different frequencies simultaneously (82 kHz, 210 kHz, and 1.57 MHz).
Flow cytometry miniaturization with Chemyx syringe pumps.
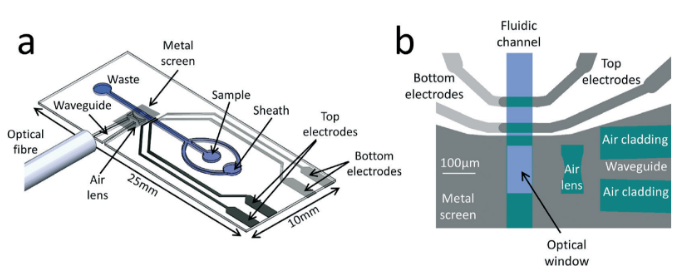
The miniaturization of flow cytometry is possible through the integration of impedance, fluorescence, and side scatter into a new platform that is portable and low-cost. Chemyx Fusion 200 loads the samples and pushes through a chip at a constant flow rate; such a reliable pump allows a ratio variation between the sample and sheath flow at a continuous 80 μL/min. Statistical analysis shows that the system is accurate and will improve the identification and quantification of different cell types as the CD4+ and the T-cell enumeration which was successfully demonstrated2.
Chemyx syringe pumps can provide a reliable operation for the development of impedance techniques such as cell counting and identification. Microfluidic operation is required in these conditions; the studies mentioned above show that is possible to miniaturize and integrate flow cytometry with impedance tools. This novel technology is currently developed with Chemyx syringe pumps, other exciting tools are discussed on the Chemyx website.
Concluding remarks
Chemyx syringe pump systems can provide a reliable operation for the development of impedance techniques such as cell counting and identification. Microfluidic operation is required in these conditions; the studies mentioned above show that is possible to miniaturize and integrate flow cytometry with impedance tools. This novel technology is currently developed with Chemyx syringe pumps, other exciting tools are discussed on the Chemyx website.
References
- Kirkegaard, J., Clausen, C.H., Rodriguez-Trujillo, R., Svendsen, W.E., 2014. Study of paclitaxel-treated HeLa cells by differential electrical impedance flow cytometry. Biosensors 4, 257–272. https://doi.org/10.3390/bios4030257
- Spencer, D., Elliott, G., Morgan, H., 2014. A sheath-less combined optical and impedance micro-cytometer. Lab Chip 14, 3064–3073. https://doi.org/10.1039/c4lc00224e

